Adsorption of volatile polonium and bismuth species on metals in various gas atmospheres: Part I - Adsorption of volatile polonium and bismuth on gold
Abstract
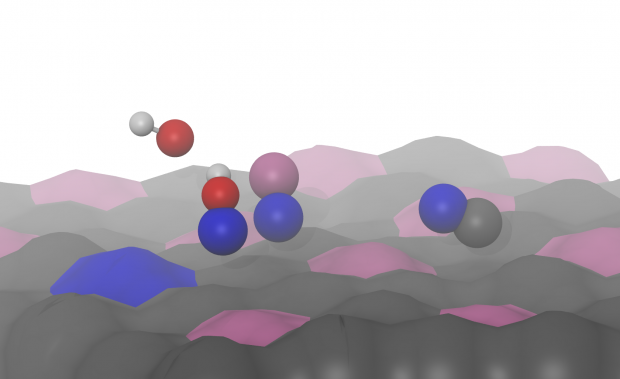
Polonium isotopes are considered the most hazardous radionuclides produced during the operation of accelerator driven systems (ADS) when lead-bismuth eutectic (LBE) is used as the reactor coolant and as the spallation target material. In this work the use of gold surfaces for capturing polonium from the cover gas of the ADS reactor was studied by thermochromatography. The results show that gaseous monoatomic polonium, formed in dry hydrogen, is adsorbed on gold at 1058 K. Its adsorption enthalpy was calculated as -250 +/- 7 kJ mol(-1), using a Monte Carlo simulation code. Highly volatile polonium species that were observed in similar experiments in fused silica columns in the presence of moisture in both inert and reducing gas were not detected in the experiments studying adsorption on gold surfaces. PoO2 is formed in both dry and moist oxygen, and its interaction with gold is characterized by transport reactions. The interaction of bismuth, present in large amounts in the atmosphere of the ADS, with gold was also evaluated. It was found that bismuth has a higher affinity for gold, compared to polonium, in an inert, reducing, and oxidizing atmosphere. This fact must be considered when using gold as a material for filtering polonium in the cover gas of ADS.

 Open Access version available at
Open Access version available at