S. Biswas, D.E.P. Vanpoucke, T. Verstraelen, M. Vandichel, S. Couck, K. Leus, Y-Y Liu, M. Waroquier, V. Van Speybroeck, J.F.M. Denayer, P. Van der Voort
Journal of Physical Chemistry C
Abstract
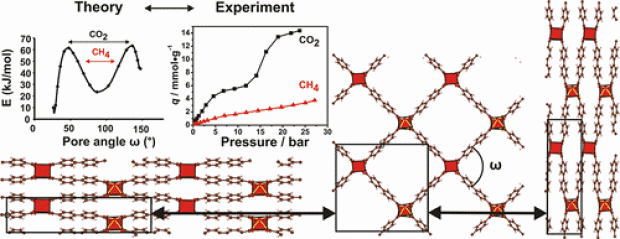
Six new functionalized vanadium hydroxo terephthalates [VIII(OH)(BDC-X)]•n(guests) (MIL-47(VIII)-X-AS) (BDC = 1,4-benzenedicarboxylate; X = -Cl; -Br, -CH3, -CF3, -OH, -OCH3; AS = as-synthesized) along with the parent MIL-47 were synthesized under rapid microwave-assisted hydrothermal conditions (170 ºC, 30 min, 150 W). The unreacted H2BDC-X and/or occluded solvent molecules can be removed by thermal activation under vacuum leading to the empty-pore forms of the title compounds (MIL-47(VIV)-X). Except pristine MIL-47 (+III oxidation state), the vanadium atoms in all the evacuated functionalized solids stayed in +IV oxidation state. The phase purity of the compounds was ascertained by X-ray powder diffraction (XRPD), diffuse reflectance infrared Fourier transform (DRIFT) spectroscopy, Raman, thermogravimetric (TG), and elemental analysis. The structural similarity of the filled and empty-pore forms of the functionalized compounds with the respective forms of parent MIL-47 was verified by cell parameter determination from XRPD data. TGA and temperature-dependent XRPD (TDXRPD) experiments in air atmosphere indicate high thermal stability in the range 330-385 ºC. All the thermally activated compounds exhibit significant microporosity (SLangmuir in the range 418-1104 m2 g-1), as verified by the N2 and CO2 sorption analysis. Among the six functionalized compounds, MIL-47(VIV)-OCH3 shows the highest CO2 uptake, demonstrating the determining role of functional groups on the CO2 sorption behaviour. For this compound and pristine MIL-47(VIV), Widom particle insertion simulations were performed based on ab initio calculated crystal structures. The theoretical Henry coefficients show a good agreement with the experimental values, and calculated isosurfaces for the local excess chemical potential indicate the enhanced CO2 affinity is due to two effects: (i) the interaction between the methoxy group and CO2 and (ii) the collapse of the MIL-47(VIV)-OCH3 framework.

 Open Access version available at UGent repository
Open Access version available at UGent repository