Abstract
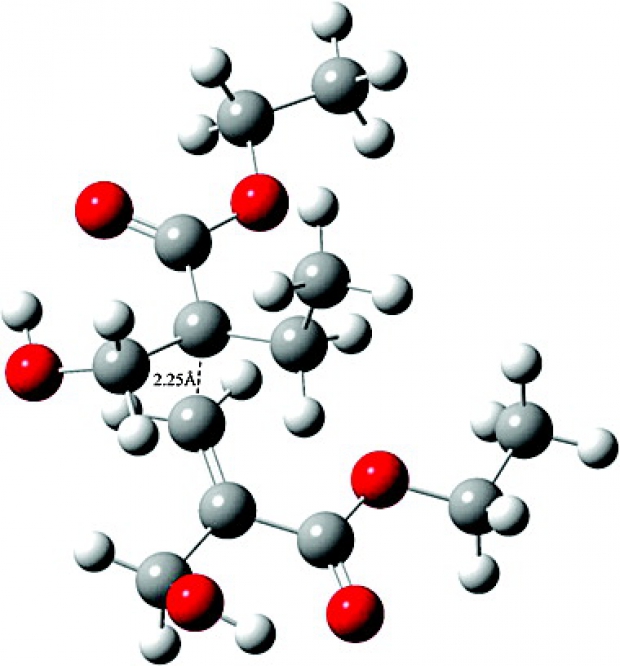
Radical polymerization processes occur through a complex network of many different reactions. It is well-known that the polymerization rate is directly related to the monomer structure. The experimental polymerizability behavior is expressed as kp/kt1/2, where kp is the rate coefficient of propagation and kt is the rate coefficient of termination. In this study, the reactivity of a series of acrylates and methacrylates is modeled in order to understand the effect of the pendant group size, the polarity of a pendant group, and the nature of the pendant group (linear vs cyclic) on their polymerizability. The geometries and frequencies are calculated with the B3LYP/6-31+G(d) methodology whereas the energetics and kinetics of these monomers have been studied using the two-component BMK/6-311+G(3df,2p)//B3LYP/6-31+G(d) level of theory. For rotations about forming/breaking bonds in the transition state, an uncoupled scheme for internal rotations has been applied with potentials determined at the B3LYP/6-31+G(d) level. Generally the rate constants for propagation kp mimic the qualitative polymerization trend of the monomers modeled and can be used with confidence in predicting the polymerizability behavior of acrylates. However in the case of 2-dimethylaminoethyl acrylate, chain transfer is found to play a major role in inhibiting the polymerization. On the other hand, the disproportionation reaction turns out to be too slow to be taken into consideration as a termination reaction.
