S. Stankovic
A difference between the pi-electron properties of catafusenes and perifusenes
On the cycle-dependence of topological resonance energy
Partitioning of pi-electrons in rings of diaza-derivatives of acenes
Solvent-controlled selective transformation of 2-Bromomethyl-2-methylaziridines to functionalized aziridines and azetidines
Abstract
The reactivity of 2-bromomethyl-2-methylaziridines toward oxygen, sulfur, and carbon nucleophiles in different solvent systems was investigated. Remarkably, the choice of the solvent has a profound influence on the reaction outcome, enabling the selective formation of either functionalized aziridines in dimethylformamide (through direct bromide displacement) or azetidines in acetonitrile (through rearrangement via a bicyclic aziridinium intermediate). In addition, the experimentally observed solvent-dependent behavior of 2-bromomethyl-2-methylaziridines was further supported by means of DFT calculations.
 Open Access version available at UGent repository
Open Access version available at UGent repositorySystematic Study of Halide-Induced Ring Opening of 2-Substituted Aziridinium Salts and Theoretical Rationalization of the Reaction Pathways
Abstract
The ring-opening reactions of 2-alkyl-substituted 1,1-bis(arylmethyl)- and 1-methyl-1-(1-phenylethyl)aziridinium salts with fluoride, chloride, bromide and iodide in acetonitrile have been evaluated for the first time in a systematic way. The reactions with fluoride afforded regioisomeric mixtures of primary and secondary fluorides, whereas secondary β-chloro, β-bromo and β-iodo amines were obtained as the sole reaction products from the corresponding halides by regiospecific ring opening at the substituted position. Both experimental and computational results revealed that the reaction outcomes in the cases of chloride, bromide and iodide were dictated by product stability through thermodynamic control involving rearrangement of the initially formed primary halides to the more stable secondary halides. The ring opening of the same aziridinium salts with fluoride, however, was shown to be mediated by steric interactions (kinetic control), with the corresponding primary β-fluoro amines being obtained as the main reaction products. Only for 2-acylaziridinium ions was the reaction outcome shown to be under full substrate control, affording secondary β-fluoro, β-chloro, β-bromo and β-iodo amines through exclusive attack at the activated α-carbonyl carbon atom.
 Open Access version available at UGent repository
Open Access version available at UGent repositorySynthesis of 3-Methoxyazetidines via an Aziridine to Azetidine Rearrangement and Theoretical Rationalization of the Reaction Mechanism
Abstract
The synthetic utility of N-alkylidene-(2,3-dibromo-2-methylpropyl)amines and N-(2,3-dibromo-2-methylpropylidene)benzylamines was demonstrated by the unexpected synthesis of 3-methoxy-3-methylazetidines upon treatment with sodium borohydride in methanol under reflux through a rare aziridine to azetidine rearrangement. These findings stand in contrast to the known reactivity of the closely related N-alkylidene-(2,3-dibromopropyl)amines, which are easily converted into 2-(bromomethyl)aziridines under the same reaction conditions. A thorough insight into the reaction mechanism was provided by both experimental study and theoretical rationalization.
Regioselectivity in the ring opening of non-activated aziridines
Abstract
In this critical review, the ring opening of non-activated 2-substituted aziridines via intermediate aziridinium salts will be dealt with. Emphasis will be put on the relationship between the observed regioselectivity and inherent structural features such as the nature of the C2 aziridine substituent and the nature of the electrophile and the nucleophile. This overview should allow chemists to gain insight into the factors governing the regioselectivity in aziridinium ring openings (81 references).
Reactivity of Activated versus Nonactivated 2-(Bromomethyl)aziridines with respect to Sodium Methoxide: a Combined Computational and Experimental Study
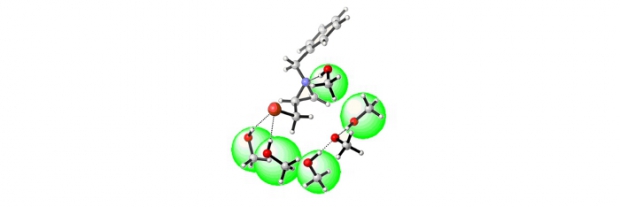
Abstract
The difference in reactivity between the activated 2-bromomethyl-1-tosylaziridine and the non-activated 1-benzyl-2-(bromomethyl)aziridine with respect to sodium methoxide was analyzed by means of DFT calculations within the supermolecule approach, taking into account explicit solvent molecules. In addition, the reactivity of epibromohydrin with regard to sodium methoxide was assessed as well. The barriers for direct displacement of bromide by methoxide in methanol are comparable for all three heterocyclic species under study. However, ring opening was found to be only feasible for the epoxide and the activated aziridine, and not for the non-activated aziridine. According to these computational analyses, the synthesis of chiral 2-substituted 1-tosylaziridines can take place with inversion (through ring opening/ring closure) or retention (through direct bromide displacement) of configuration upon treatment of the corresponding 2-(bromomethyl)aziridines with one equivalent of a nucleophile, whereas chiral 2-substituted 1-benzylaziridines are selectively obtained with retention of configuration (via direct bromide displacement). Furthermore, the computational results showed that explicit accounting for solvent molecules is required to describe the free energy profile correctly. To verify the computational findings experimentally, chiral 1-benzyl-2-(bromomethyl)aziridines and 2-bromomethyl-1-tosylaziridines were treated with sodium methoxide in methanol. The presented work concerning the reactivity of 2-bromomethyl-1-tosylaziridine stands in contrast to the behaviour of the corresponding 1-tosyl-2-(tosyloxymethyl)aziridine with respect to nucleophiles, which undergoes a clean ring-opening/ring-closure process with inversion of configuration at the asymmetric aziridine carbon atom.
