Reductive imino-pinacol coupling reaction of halogenated aromatic imines and iminium ions catalyzed by precious metal catalysts using hydrogen
Abstract
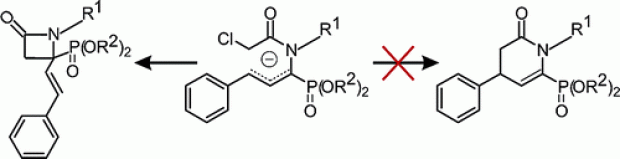
The first heterogeneously catalyzed process for the reductive coupling of imines and iminium ions is reported using precious metal catalysts in combination with hydrogen gas as the terminal reductant. The optimized method in terms of catalyst composition and reaction conditions allowed to produce aromatic vicinal diamines without the use of stoichiometric amounts of zero or low valent metals, which is currently the preferred method. The most important mechanistic features of the reaction were unraveled by a combined experimental and computational approach. The developed methodology is very efficient for the coupling of aromatic iminium ions with yields up to 88 % while imines give only low to moderate yields.