P. Ramaswamy, J. Wieme, E. Alvarez, L. Vanduyfhuys, J.-P. Itié, P. Fabry, V. Van Speybroeck, C. Serre, P.G. Yot, G. Maurin
Journal of Materials Chemistry A
Abstract
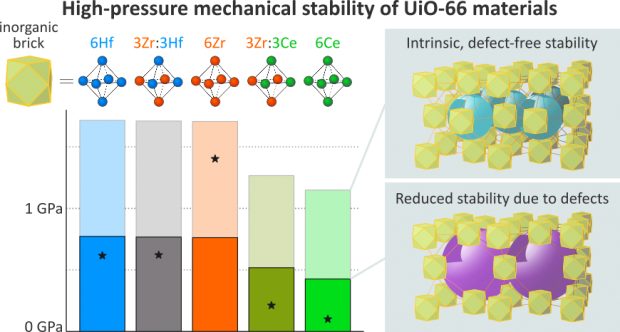
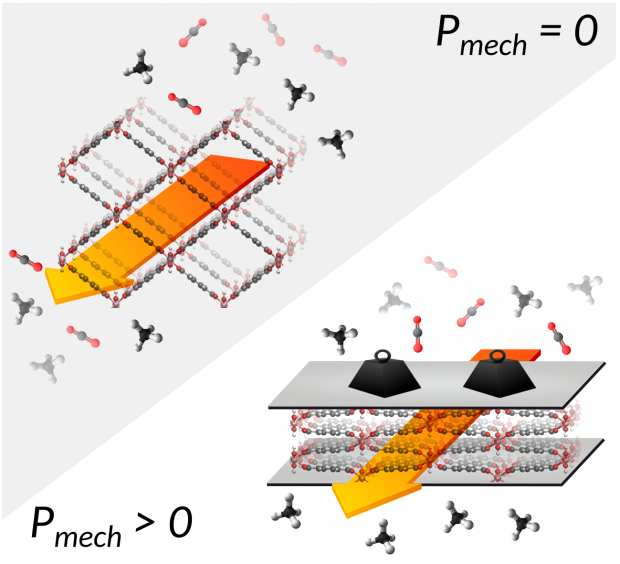
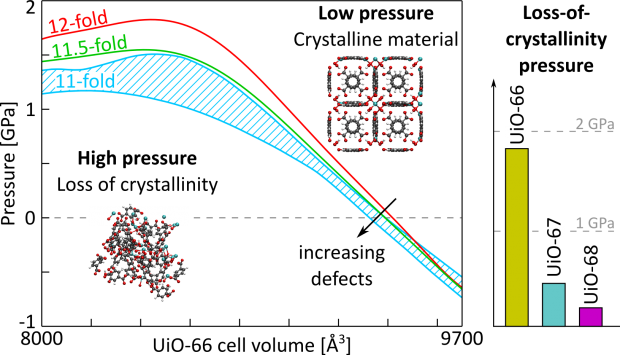
A gallium analogue of the commercially available Al-fumarate MOF A520 - recently identified as isotypic to MIL-53(Al)-BDC - has been synthesized and further characterized in its hydrated and dehydrated forms. The structural response under applied mechanical pressure of this MIL-53(Ga)-FA solid was investigated using advanced experimental techniques coupled with computational tools. Hg porosimetry and high-pressure X-Ray Powder Diffraction (XRPD) experiments evidenced that the pristine dehydrated large pore form undergoes an irreversible structure contraction upon an applied pressure of 85 MPa with an associated volume change of ca. 14% which makes this material promising for mechanical energy storage applications, in particular as a shock absorber. The breathing behavior was further rationalized performing a series of periodic Density Functional Theory (DFT) calculations with the construction of an energy profile as a function of volume for both MIL-53(Ga)-FA and its Aluminum analogue. As such we could fully unravel the microscopic origin of the difference in pressure-induced behavior for the aluminum and gallium fumarate based materials.

 Open Access version available at
Open Access version available at