Orthogonal electronic structure engineering in nanoporous materials for the design of new-generation photocatalysts for clean energy conversion.
Orthogonal electronic structure engineering in nanoporous materials for the design of new-generation photocatalysts for clean energy conversion.
Promotor(en): V. Van Speybroeck, P. Van der Voort /28148 / Nanoporous materials, SpectroscopyBackground and problem
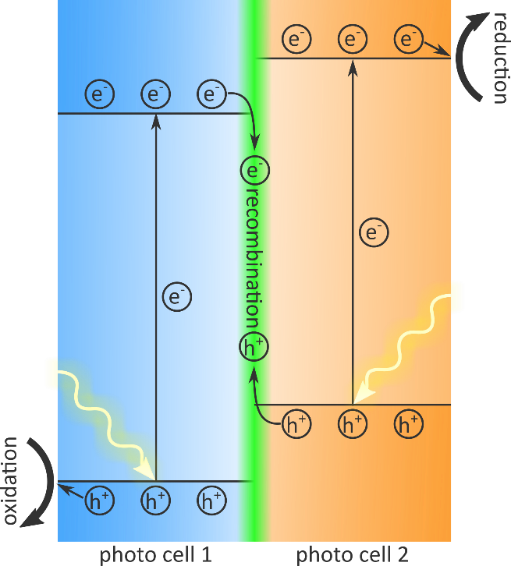
Sunlight is abundantly present and freely available as an energy source. Therefore, the design of photoactive devices that efficiently harness sunlight is a key aspect in the transition to a sustainable economy. The harvested solar energy can, for example, be used in electrochemical reactions which generate hydrogen gas through water splitting, or reduce carbon dioxide. The efficiency of a photoactive device can be characterized by three processes: (i) the absorption of photons to generate electron/hole pairs, (ii) the separation and migration of these charge carriers, and, for photocatalysis, (iii) the chemical oxidation and reduction mediated by these charge carriers. The simultaneous optimization of these three processes requires specialized photocatalytic systems and materials. A promising approach that has recently attracted much attention is the use of a so-called Z-scheme (Figure 1) [1], which requires two materials with a well-tuned bandgap. Both materials absorb sunlight efficiently to create holes in the valence band and electrons in the conduction band. For one material, the ionization energy is high enough such that it can donate holes to the oxidation reaction, while for the other the electron affinity is low enough such that it can donate electrons to the reduction reaction. The Z-scheme is completed when the electrons of the first material recombine with the holes of the second, owing to the alignment of the energy levels of valence and conduction bands for both materials.
A potentially viable class of materials, as indicated by our experimental partners, are covalent organic frameworks (COFs) [2]. These are crystalline materials that are assembled by organic building blocks and linked by strong covalent bonds into a framework structure, which can extend in all three dimensions (3D COFs) or only in two dimensions (2D COFs), which gives rise to a graphite like structure (see Figure 2). As their physicochemical properties, such as the bandgap, can be tuned on a molecular level by carefully selecting the appropriate building blocks, they are tractable for a variety of applications. For Z-scheme systems, the focus lies on the layered 2D COFs as they have an extended conjugated π-network, ensuring fast charge transport, ideal for our purposes.

As the number of COF building blocks grows rapidly, experimental characterization of each possible structure is an impossible task. A computational screening of the material space forms a valuable alternative and can guide experimental efforts by predicting promising 2D COFs that can be combined into an efficient Z-scheme. In recent years, the Center for Molecular Modeling (CMM) has developed various computational tools and workflows to both create and screen large databases of building block materials [3]. However, a screening of electronic properties has not been achieved yet, as these require heavy calculations for each combination of building blocks to reliably predict the electronic structure, which significantly limits the predictive power of our toolbox. An alternative approach, referred to as orthogonal electronic structure engineering (OESE), states that the electronic structure of building block materials can be deduced via decoupled contributions of their individual building blocks. While pioneering work on metal-organic frameworks (MOFs) [4-6] and a limited set of COFs [7] show large promise, the OESE principle has not yet been applied on a large dataset, which would reliably bridge the gap between our screening software and industrial electronic structure requirements.
Goal
The overall goal of this thesis is to propose a set of COFs that can be adopted by our experimental partner in an efficient Z-scheme system, following a two-step approach. In the first step, the student will become familiar with the basics of electronic structure calculations and the OESE principle. It is the aim to gain insight into the overall applicability and the limitations of the OESE principle for 2D COFs, which will provide the necessary background to construct a reliable workflow for calculating decoupled electronic structure contributions and predicting the electronic structure of the periodic material through density functional theory. The reliability of this workflow will be critically assessed by considering the commensurability between the electronic structure of the isolated building block and the framework for certain well-known 2D COFs. In particular, it will be crucial to investigate the influence of the manner in which the isolated building blocks are defined and their specific embedding in the periodic framework. The student will benefit from the expertise present at the CMM in performing electronic structure calculations and employing the OESE principle [6-7].
In the second part of the thesis, the OESE principle will be applied on a broader scale to identify a set of COFs that are suited for industrial application in a Z-scheme system. The electronic structure will be calculated for a library of building blocks, and the absolute energy levels of the valence and conduction bands for all possible combinations will be derived to determine the optimal COF materials to use in a Z-scheme. A library of COF building blocks is already established at the CMM, but can be extended using building blocks proposed by our experimental partner, i.e. the Centre for Ordered Materials, Organometallics and Catalysis (COMOC) in Ghent.
The student will be actively coached to make him/her acquainted with the electronic structure calculation techniques early in the thesis year, and to transfer necessary programming skills needed to perform the research. Furthermore, the student could be involved in the running project with our experimental partners, which can experimentally synthesize the proposed COFs to validate their efficacy as Z-scheme materials.
- Study programmeMaster of Science in Engineering Physics [EMPHYS], Master of Science in Physics and Astronomy [CMFYST]Keywordsdensity of states, electronic structure coupling, Covalent organic frameworks, Z-schemeReferences
[1] Quanlong Xu, Liuyang Zhang, Jiaguo Yu, Swelm Wageh, Ahmed A. Al-Ghamdi, Mietek Jaroniec, "Direct Z-scheme photocatalysts: Principles, synthesis, and applications," Materials Today, Volume 21, Issue 10, pp. 1042-1063, 2018.
[2] A. P. Côté, A. I. Benin, N. W. Ockwig, M. O'Keeffe, A. J. Matzger and O. M. Yaghi, “Porous, crystalline, covalent organic frameworks,” Science, vol. 310, no. 5751, pp. 1166-1170, 2005.
[3] S. Borgmans, S. M. J. Rogge, J. S. De Vos, C. V. Stevens, P. Van Der Voort and V. Van Speybroeck, “Quantifying the likelihood of structural models through a dynamically enhanced powder X-ray diffraction protocol,” Angewandte Chemie International Edition, 2021. DOI: 10.1002/anie.202017153.
[4] R. Grau-Crespo, A. Aziz, A. W. Collins, R. Crespo-Otero, N. C. Hernández, L. M. Rodriguez-Albelo, A. R. Ruiz-Salvador, S. Calero en S. Hamad, “Modelling a linker mix-and-match approach for controlling the optical excitation gaps and band alignment of zeolitic imidazolate frameworks,” Angewandte Chemie, vol. 128, nr. 52, pp. 16246-16250, 2016.
[5] M. A. Syzgantseva, C. P. Ireland, F. M. Ebrahim, B. Smit en O. A. Syzgantseva, “Metal Substitution as the Method of Modifying Electronic Structure of Metal-Organic Frameworks,” Journal of the American Chemical Society, vol. 141, nr. 15, pp. 6271-6278, 2019.
[6] A. De Vos, K. Hendrickx, P. Van Der Voort, V. Van Speybroeck en K. Lejaeghere, “Missing linkers: an alternative pathway to UiO-66 electronic structure engineering,” Chemistry of Materials, vol. 29, nr. 7, pp. 3006-3019, 2017.
[7] A. De Vos, K. Lejaeghere, F. M. Miranda, C. V. Stevens, P. Van Der Voort en V. Van Speybroeck, “Electronic properties of heterogenized Ru (ii) polypyridyl photoredox complexes on covalent triazine frameworks,” Journal of Materials Chemistry A, vol. 7, nr. 14, pp. 8433-8442, 2019.
