Diffusion channels for small molecules in nanoporous systems with blocked sites
Diffusion channels for small molecules in nanoporous systems with blocked sites
Promotor(en): V. Van Speybroeck, A. Ghysels /15_NANO13 / Nanoporous materialsNanoporous materials are structurally ordered materials that possess channels and pores of molecular dimensions, which makes them very tractable for various applications such as separation processes, gas storage, ... Zeolites are omnipresent in a wide variations of industrial applications. The materials are tecto-silicates, built out of SiO4 and AlO4 tetrahedra linked into corner-sharing networks; and when aluminium is present in the framework, electro-neutrality requires the incorporation of charge compensating cations, which occupy extra-framework sites in the channels and cages. The function of these materials relies to a large extent on the diffusion of small molecules through the channel and pore system as schematically shown in Figure 1.
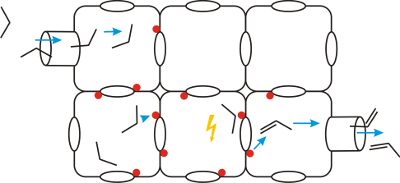
Figure 1: Schematic representation of various paths of small molecules through the pore and channel system of nanoporous materials.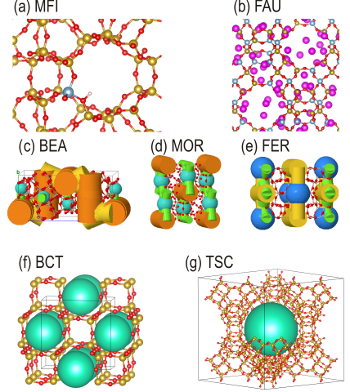
Figure 2: Some example zeolite structures with different architectures and channel system.The easiness of these diffusion paths relies on the shape and dimensions of the channel and pore framework (Figure 2). Molecular simulations have contributed to a great extent to the molecular level understanding of diffusion paths in naporous materials.
Diffusion is a phenomenon that takes place at larger length and time scales. It is typically studied using classical force field based approaches. In molecular dynamics simulations the position of the molecule is tracked over time, from which the diffusivity can be derived, as shown for propene in Figure 3. In some cases, molecules have to surmount energy barriers during their paths through the crystal. Diffusion then becomes a rare event, and special techniques are needed to describe the hopping process from one cage to the other. Furthermore in a variety of important cases, blocked sites are present in the pores of the material such as organic fragments. These species block the channel system and restrict the diffusion. A proper understanding of diffusion is mandatory for understanding molecular channels in nanoporous materials, which play a vital role in many applications.
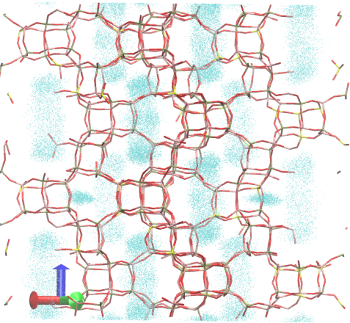
Figure 3: Trajectories of the diffusion of propylene at 300K in H-SAPO-56. Each dot represents the position of a propene molecule at a certain time step. The total simulation time amounts here to 10 ns.Goal In the framework of this master thesis, we want to study the diffusion of small molecules such as ethene and propene through various industrially relevant zeolites. We wish to study the diffusion in cases where blocked sites are available in the pores of the system. Despite the wealth of literature available on the topic these processes have not been studied to date. The work will be performed using in-house developed software codes to perform molecular dynamics simulations and if necessary also Monte Carlo simulations. The potential energy surface will be described using classical force fields which are available from literature. The final intention is to understand the factors that control the diffusion in zeolites and to assign the molecular factors contributing to molecular channel systems.
Once blocked sites are available in the pores of the material, it will no longer be possible to use the available software tools to study the diffusion process. In that case new advanced molecular dynamics techniques will have to be implemented in our in-house developed software codes to construct the free energy surface for these so-called rare events. The CMM has ample experience in modelling nanoporous materials, has developed over the years various methods to study phenomena at the nanoscale from statistical physics and from thermodynamics while relying on molecular input.
The proposed research is part of a larger concerted action in which physical and chemical transformations are studied at the nanoscale within nanoporous materials. In that framework the CMM collaborates with leading experimental and theoretical partners. It is the intention to involve the interested student in our workdiscussions with our collaborators.
- Study programmeMaster of Science in Engineering Physics [EMPHYS], Master of Science in Chemical Engineering [EMCHEM], Master of Science in Physics and Astronomy [CMFYST]ClustersFor Engineering Physics students, this thesis is closely related to the cluster(s) nano, modelingKeywordsDiffusion, Molecular dynamics simulations, Nanoporous materials, Molecular sievesRecommended coursesSimulations and modeling for the nanoscale, Moleculaire modellering van industriele processen, Computational physics



