From a dense to a sponge-like structure at high pressures: unravelling the counter-intuitive behaviour of zinc cyanide
From a dense to a sponge-like structure at high pressures: unravelling the counter-intuitive behaviour of zinc cyanide
Promotor(en): V. Van Speybroeck /17NANO10 / Nanoporous materialsFramework materials are not your everyday type of solids. They are entirely different from typical metals or ceramics, which all have one thing in common: at the atomic scale, they correspond to closely packed crystals. In contrast, framework materials have large cavities inside them (also called pores), which make them attractive for many applications. Pores allow confining molecules, for example, making them ideally suited to store gases, catalyse chemical reactions or act as biomedical vehicles for drugs transport. In addition, the porosity of framework materials can be switched on or off by means of external triggers. There are materials that become porous by absorbing light or by adsorbing particles. At high pressure, most framework materials react in the same way, however, by collapsing to a denser phase.
In this respect, zinc cyanide is a very odd material. Researchers recently discovered that this molecular crystal does exactly the opposite of other framework materials: at sufficiently high pressures, and using the right pressure-inducing medium, pores are generated in the originally dense structure! This strange behaviour already starts when compressing the Zn(CN)2 crystal slightly. Whereas normal materials become harder as you push more, zinc cyanide becomes softer instead. At pressures around 1.5 GPa, the material undergoes a phase transformation, and the resulting phase depends on the pressure-inducing medium. When you use a liquid with large molecules, the expected reaction takes place, and the Zn(CN)2 structure becomes even denser. When using water, methanol or a mixture of methanol, ethanol and water, however, three different high-pressure phases can be obtained, each having a larger volume – with pores – than the original material. This strange behaviour is depicted in the figure. The increase in volume is due to the interaction between zinc cyanide and the pressure-transmitting molecules, which enter the framework and push the pores open. Although the volume of zinc cyanide increases in this way, the combined volume of zinc cyanide and pressure-inducing medium still becomes smaller, so thermodynamic laws are still satisfied.
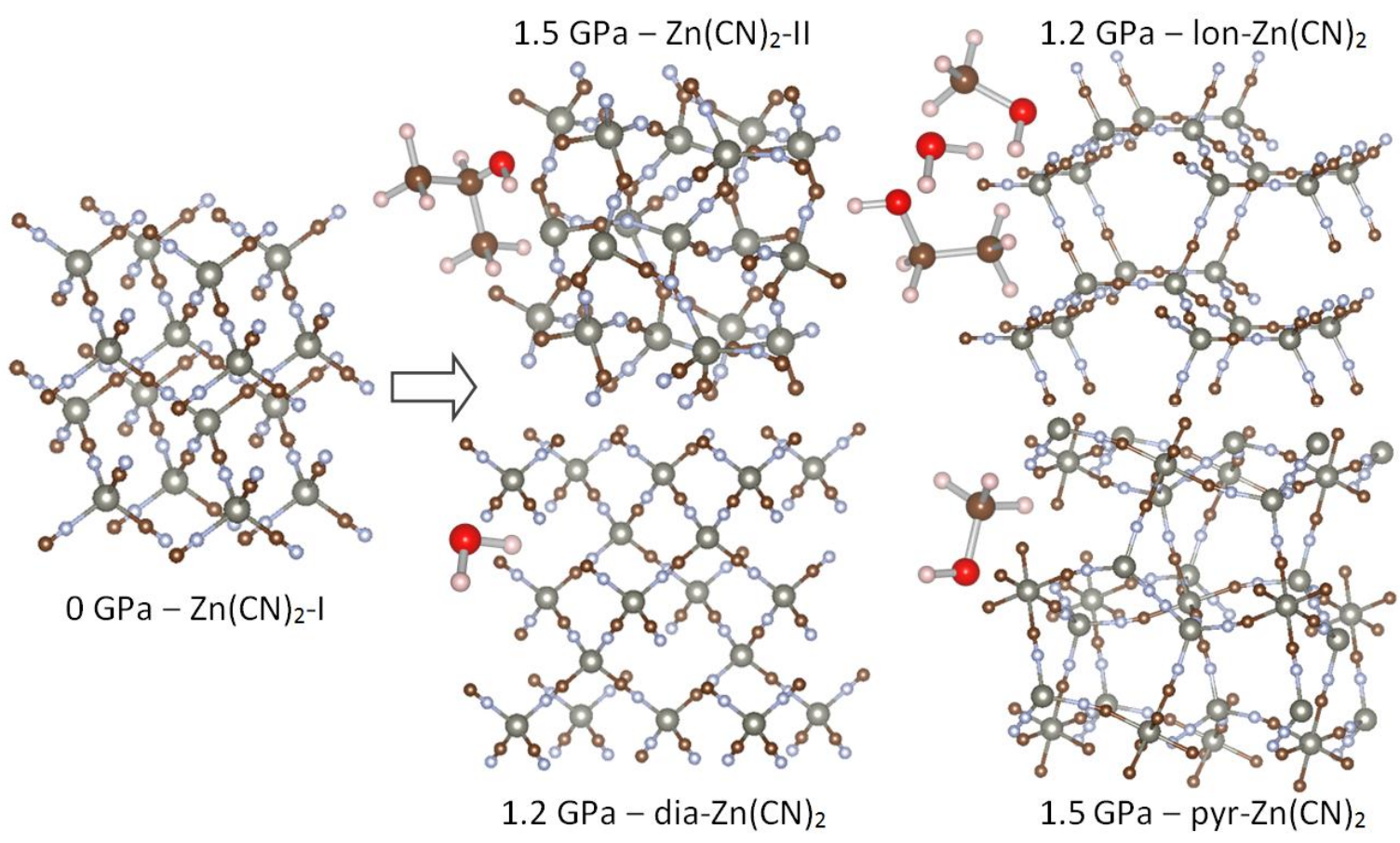
The counter-intuitive reaction of zinc cyanide to pressure can be very useful. Not only is it now possible to store molecules in this framework material, but it can also be used as a sensor to pressure, with the pores opening up as soon as a certain threshold pressure is reached. If we were able to understand how the zinc cyanide interacts with different adsorbed molecules, and how pressure affects the stability of the different Zn(CN)2 phases, this knowledge could be used to create pores in other promising materials.
Goal
This Master's thesis aims to investigate the deformation behaviour of zinc cyanide and its high-pressure phases, and unravel the role of adsorbed molecules. To do this, you will use density-functional theory (DFT), a method based on quantum physics and implemented in the VASP program. Ab initio molecular dynamics, as implemented in the CP2K program, may moreover be used to take into account the effect of temperature.
First, you will test whether you are able to reproduce experimentally measured properties of adsorbate-free zinc cyanide. It is still not clear how the pressure-induced softening of zinc cyanide depends on temperature, so this is a particularly interesting question to investigate as well. When this validation has succeeded, you will have a closer look at the high-pressure phases of Zn(CN)2, on which even less is known. You will determine the mechanical and energetic stability of these structures under pressure, both with and without adsorbates, and investigate how the particular choice of adsorbate influences which high-pressure phase is obtained. You will also have a closer look at the role of disorder in this process: it was experimentally found that some zinc cyanide structures have a variable number of carbon and nitrogen atoms around the zinc centres, which might affect their high-pressure properties both with and without the presence of guest molecules.
Context for Engineering Physics students
Physics: use of quantum mechanical models for materials modelling.
Engineering: application to the materials properties of molecular frameworks.
- Study programmeMaster of Science in Engineering Physics [EMPHYS], Master of Science in Physics and Astronomy [CMFYST]ClustersFor Engineering Physics students, this thesis is closely related to the cluster(s) MODELLING, MATERIALS, NANOKeywordszinc cyanide, pressure-induced softening, Adsorption, density-functional theoryReferences
S.H. Lapidus et al., 'Exploiting High Pressures to Generate Porosity, Polymorphism, And Lattice Expansion in Nonporous Molecular Framework Zn(CN)2', J. Am. Chem. Soc. 135, 7621-7628 (2013).

