Active site engineering of zeolite catalysts for alkene cracking through metal incorporation
Active site engineering of zeolite catalysts for alkene cracking through metal incorporation
Promotor(en): V. Van Speybroeck /20NANO01 / Nanoporous materialsProbleemstelling:
Zeolite catalyzed alkene cracking plays a crucial role in both classical petrochemical processes, like FCC, as well as emerging alternatives, such as the methanol-to-olefins (MTO) process or biomass conversion. Recent trends like the increased shale gas production and the necessity for global CO2 emission reductions have a serious impact on the production of base chemicals such as ethene and propene. On-purpose production processes of light olefins, aromatics, etc. are nowadays becoming more and more important. One strategy to improve the light olefin selectivity and reduce catalyst deactivation is the modification of the zeolite active sites through incorporation of phosphorus, alkaline earth metals (Mg, Ca,…) or transition metals (Fe, Cu, Zn,…). [1-3] However, the precise influence of these modifications on the cracking mechanism is not fully understood. Nevertheless, such insight is required to select or design the optimal catalyst for catalytic cracking units.
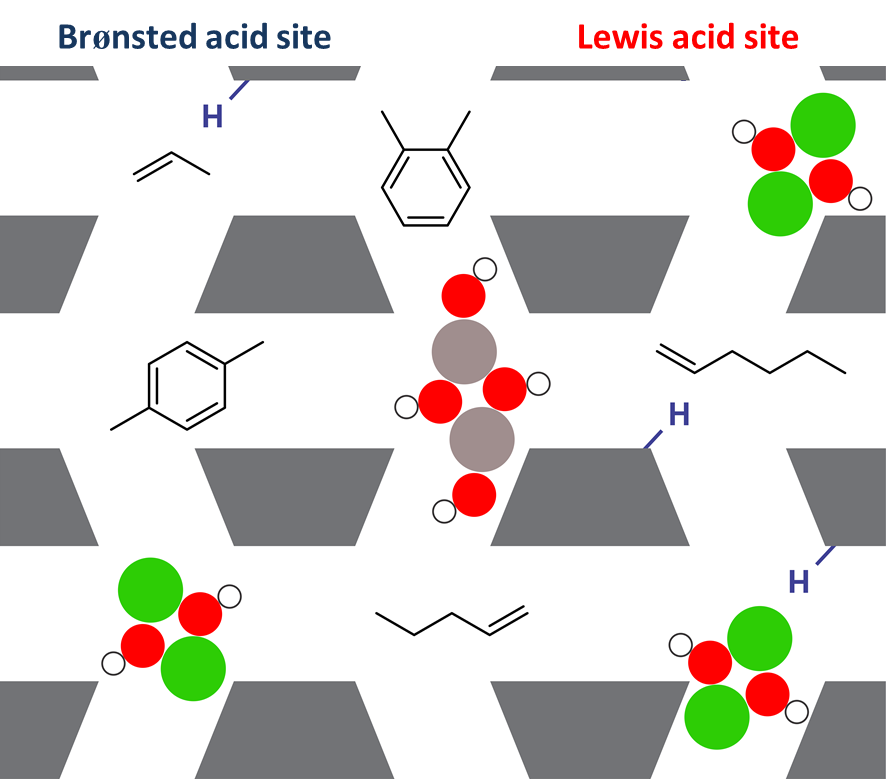
Figure 1. Schematic representation of a ZSM-5 catalyst with Brønsted acid sites (blue) and Lewis acid sites (red) originating from metal incorporation in the framework. [2]
Alkene cracking occurs through a complex reaction network of hydride transfer, isomerization, oligomerization and β-scission reactions in which carbenium ion intermediates are known to play an active role. [4] However, due to the high reactivity of these intermediates, detailed information on individual reaction steps can hardly be obtained from experiments. Ab initio simulations can assist in unraveling the reaction mechanism at the molecular level. Today, many modeling studies represent zeolites as ideal materials containing isolated Brønsted acid sites. In reality, however, zeolites are more complex and contain a combination of active sites with Lewis acidic and Brønsted acidic properties. Each of these sites may have a distinct influence on the stability of the intermediates and on the preferred cracking pathways. Advanced molecular simulations at operating conditions are essential to elucidate the impact of active site modifications on the reaction mechanism.
Doelstelling:
The goal of this master thesis is to study how active site engineering by metal incorporation in the zeolite will influence the stability and reactivity of alkene cracking intermediates. First, you will investigate the nature of the metal modified active sites in the zeolite pores. After a detailed characterization of the active sites, the second phase will consist of assessing the stability of a broad range of cracking intermediates at the various active sites. Thirdly, you will look into the effect of these modified active sites on the reaction rate of some key elementary steps (hydride transfer, oligomerization, β-scission …). To achieve this goal, you will perform a state-of-the-art molecular dynamics study which allows to mimic realistic process conditions. Ultimately, the outcome of these simulations will result in a thorough comparison of different zeolite modifications and give an answer to the question how modifications can have a beneficial effect on the catalyst selectivity and lifetime.
This project requires the application of several advanced molecular simulation techniques, which rely on first principle static and molecular dynamics simulations. The Center for Molecular Modeling has built up vast expertise in these simulation techniques and collaborates on the subject with leading experimental partners. The student will be actively coached to get acquainted with the simulation techniques needed to tackle the proposed problem. The necessary computational resources to execute this research project will be provided by the CMM. The proposed topic is challenging and requires technical skills, creativity and chemical insight.
- Study programmeMaster of Science in Chemical Engineering [EMCHEM]KeywordsHeterogeneous Catalysis, Zeolites, Catalytic Cracking, CarbocationsReferences
[1] H.E. van der Bij and B.M. Weckhuysen, Chem. Soc. Rev. 44 (2015) 7406-7428.
[2] I. Yarulina et al., Nat. Chem. 10 (2018) 804-812.
[3] E. Epelde et al., Appl. Catal. A 505 (2015) 105-115.
[4] P. Cnudde et al., ACS Catal. 8 (2018) 9579-9595.
