Unraveling methanol conversion in chabazite by computational spectroscopy and first principle chemical kinetics
Unraveling methanol conversion in chabazite by computational spectroscopy and first principle chemical kinetics
Promotor(en): V. Van Speybroeck /17SPEC05 / SpectroscopyThe conversion of methanol into hydrocarbons (MTH) or olefins (MTO) received a lot of attention during the last decades as methanol can be produced from natural gas, biomass or coal. The conversion is catalyzed by zeolite or zeotype materials. It has been found that the inorganic catalyst framework and occluded hydrocarbons catalyzed the MTO reactions. Moreover, a couple of catalytic cycles operate simultaneously during olefin formation, resulting in a very complex reaction mechanism which is currently still not fully understood. Many factors as framework topology, acid strength and process conditions determine which cycle dominates the product formation. Detailed molecular-level insights into the complex reaction network can be obtained by molecular simulations.
Both aromatics and aliphatics – the so-called hydrocarbon pool compounds – were found to play a crucial role in the formation of olefins from methanol as indicated in Figure 1. However, many theoretical studies point out that some of these cycles still exhibit very high reaction barriers, indicating that the real operating mechanism isn’t discovered yet.[1] There is an urgent quest to reveal the nature of the active routes to assist the experimentalists in tuning the catalyst towards optimal performance.
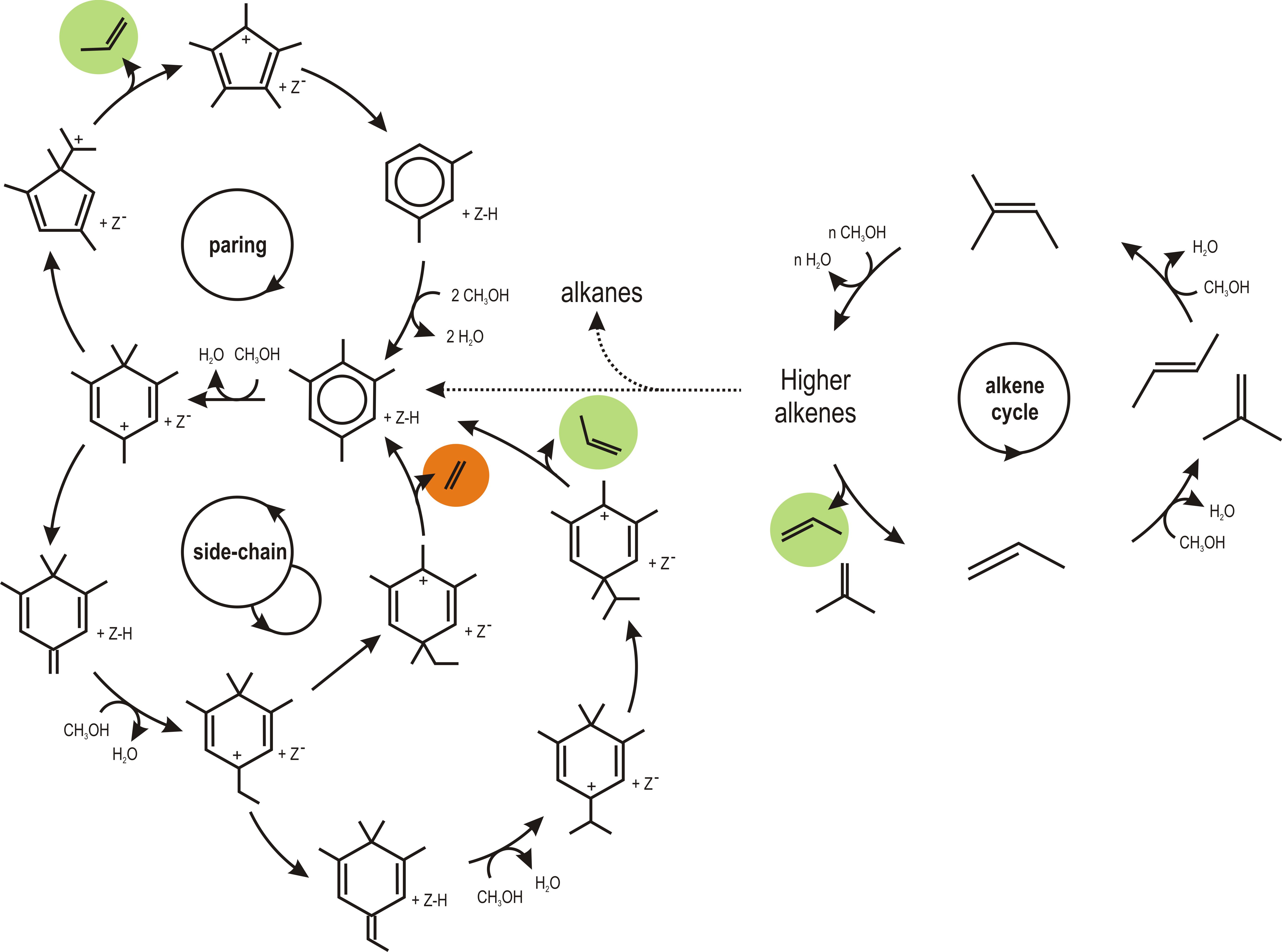
Figure 1. Overview of the proposed mechanisms for olefin formation during methanol conversion.
Doelstelling:Operando infrared and Raman spectroscopic tools give experimentalists direct access to the complex chemistry occuring in zeolites exposed to MTO conditions. However, the interpretation of the obtained spectra is often very cumbersome. For this task, computational spectroscopy can provide invaluable insights as assignment scales can be constructed for typical MTO intermediates.[2] Once characteristic peaks in experimental spectra can be uniquely assigned to MTO intermediates, they can be followed during time to extract kinetic information. In this view, the student will first construct an assigment scale that links MTO intermediates with experimental infrared and Raman spectra. In a second phase, a thorough analysis of the experimental spectroscopic data will be combined with first principle kinetic calculations to obtain a deeper understanding of the complex MTO chemistry.
The simulation of spectra and chemical kinetics requires a variety of state-of-the-art modeling techniques, for which the Center for Molecular Modeling has ample experience and can provide sufficient computational resources. Furthermore, this research will be performed in very close collaboration with an experimental partner providing the spectroscopic data. The student will also be involved in work discussions with our experimental partner. The proposed topic is challenging and requires technical skills, creativity and chemical insight.
- Study programmeMaster of Science in Chemical Engineering [EMCHEM]KeywordsHeterogeneous Catalysis, Chemical kinetics, Computational applications, SpectroscopyReferences
[1] K. Hemelsoet, J. Van der Mynsbrugge, K. De Wispelaere, M. Waroquier, V. Van Speybroeck, Unraveling Reaction Mechanisms governing Methanol-To-Olefins Catalysis Combining Theory with Experiment, ChemPhysChem, 14 (2013) 1526-1545.
[2] K. Hemelsoet, Q. Qian, T. De Meyer, K. De Wispelaere, B. De Sterck , B.M. Weckhuysen, M. Waroquier, V. Van Speybroeck, Identification of Intermediates in Zeolite-Catalyzed Reactions by In Situ UV/Vis Microspectroscopy and a Complementary Set of Molecular Simulations, Chem. Eur. J., 19 (2013) 16595-16606.

