Nanofibers for drug delivery: Modelling Polymer-Drug interaction, how to obtain the most stable yet most drug releasing formula
Nanofibers for drug delivery: Modelling Polymer-Drug interaction, how to obtain the most stable yet most drug releasing formula
Promotor(en): V. Van Speybroeck, K. De Clerck /19CHEM01 / Chemistry & BiochemistryWith over 2/3rds of all drugs in development being classified as poorly water-soluble, it can be said that the pharmaceutical oral drug delivery industry is up for a challenge. Low water-solubility results in a low bioavailability of these active pharmaceutical ingredients, APIs, rendering them useless. As the main reason for this low water-solubility is the high crystallinity of the APIs, extensive research is being put into circumventing the crystallinity by obtaining and maintaining the API in its amorphous solid state. This is generally done through the use of amorphous solid dispersions of the API in a polymeric matrix. Several techniques such as spray drying or hot melt extrusion are used today to obtain such a solid dispersion, however the biggest obstacle is its time stability. As the amorphous state is essentially a meta-stable phase, current formulations have a tendency to recrystallize over time.
Because of this reason the solvent electrospinning technique is looked at to produce such polymer-drug amorphous solid dispersions. Because of the extremely rapid solvent evaporation during the solvent electrospinning process, it is believed that the API is frozen in a random and highly homogeneous manner in the polymeric matrix. Another advantage of the solvent electrospinning technique, is that it creates nanofibrous membranes, which exhibit some unique and highly beneficial properties. The most important one being the very high specific surface, which allows for a faster dissolution rate.
Yet, one of the most important and least studied factors in making an efficient amorphous solid dispersion is selecting the right polymer for the API. In order to have an enhanced bioavailability the drug needs to remain amorphous during storage and released when swallowed. In other words, the interaction between polymer and API is a crucial parameter. Sufficient interaction is required to guarantee time stability, however the strength of the interaction should also be limited in order to allow for the highest possible API release upon swallowing.
It is very hard to obtain insight into the molecular interactions from experimental point of view. In this sense molecular modeling may provide complementary insights as this allows to gain deep molecular understanding into the interactions at working conditions, i.e. the aqueous environment, temperature, pressure and the applied polymers and API’s. Deep insights into the nanoscale interactions may guide the experiments towards a better polymer and API design. Such information on interactions also allows to enhance our chemical technological knowledge, e.g. on the meso-scale regarding multi-scale design of polymer based processes. Such information on interactions also allows to enhance our chemical technological knowledge, e.g. on the meso-scale regarding multi-scale design of polymer based processes.
Goal
The goals of this master topic is to model which part of the API flubendazole interact with biocompatible polymers. A distinction will be made between H-bonding, Van Der Waals forces, and to determine the strength of these interactions. This will be done through the model API flubendazole, which will be paired with biocompatible polymers such as poly(2-alkyl-2-oxazoline)s. The results of the models can eventually be experimentally validated.
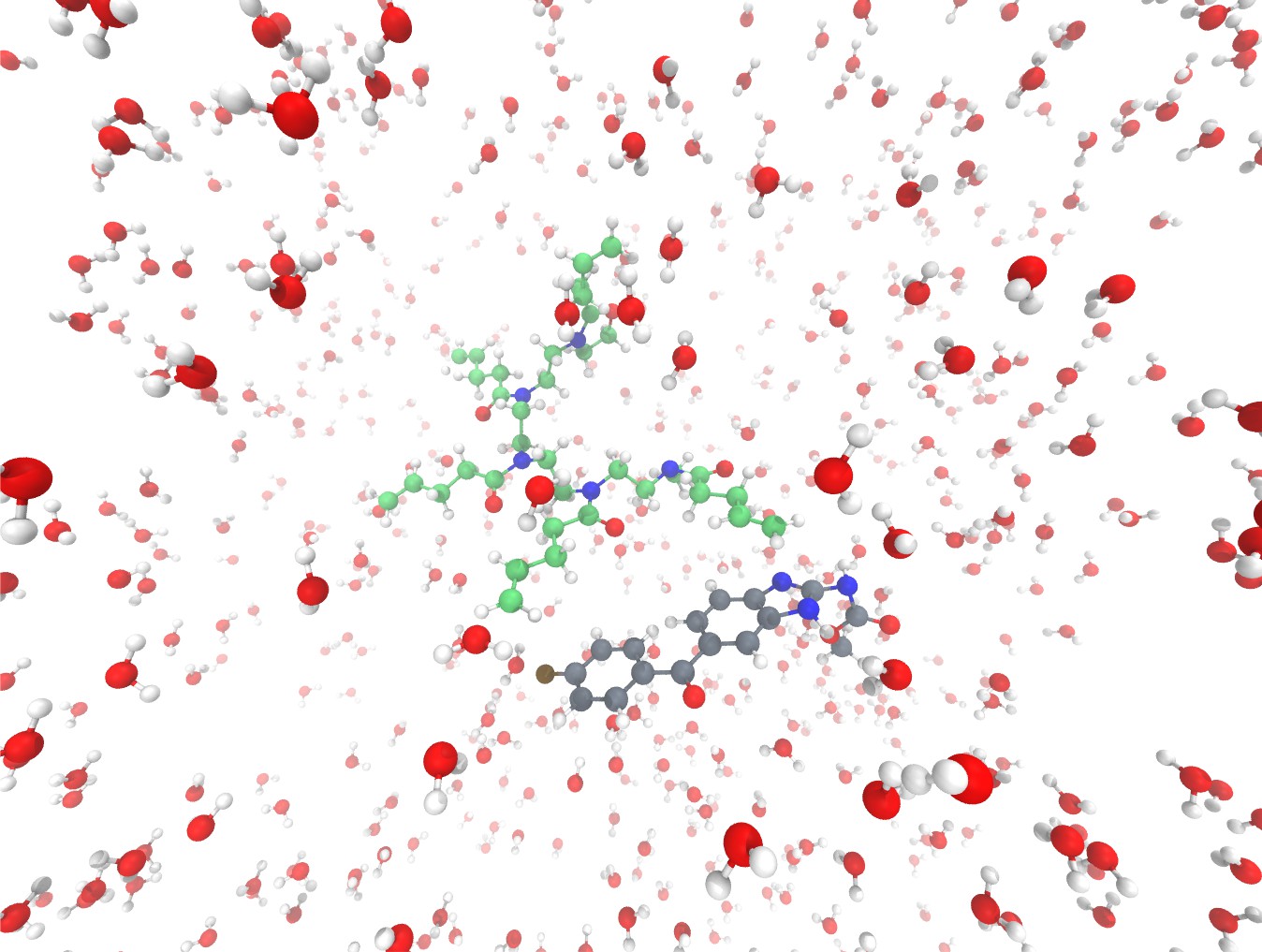
Figure 1. Example of possible interactions between a poly(2alkyl-2-oxazoline) (poly(2-but-3-enyl-2-oxazoline, green) and the model API, flubendazole (gray). In this approach the solvent environment is explicitly modelled.
To obtain these goals, a multilevel modeling approach will be followed, starting from accurate calculations at the Density functional theory (DFT) level. These techniques solve the quantum mechanical electronic structure problem and allow to distinguish between different types of interactions both inter- and intramolecular. At this level small model systems will be used for the polymers. At a next step, the system may be upscaled towards a true environment of the biocompatible polymers and water. Such calculations need to be performed using force fields which allow to follow the interactions at play at more realistic time scales, which can reach the nanosecond timescale in contrast to DFT which only allows for a few 100’s of picoseconds, while accounting for a more realistic environments.
Finally the multilevel approach will allow to obtain a deep understanding into the molecular configurations of the nanofiber, the API and solvent, to pinpoint the crucial interactions which are important in using the APIs in realistic conditions.
The calculations will be performed at the Center for molecular modeling with available software packages. The student will be actively coached to get familiar with the modeling programs. The calculations will performed on the high-performance computing facilities of the UGent and the GPU facilities of the Vrije Universiteit Brussel (VUB).
The Center for molecular modeling has already expertise in modeling poly-oxazolines.1–3 Furthermore the research group of Prof. Karen De Clerck, with its expertise in the solvent electrospinning technique, has an active collaboration with the CMM, to unravel interactions between dyes in solvents.
Aspects
Master of Science in Biomedical Engineering:
API’s are an important class of pharmaceuticals which suffer from poor solubility within the body, the use of nanofibers to enhance API uptake rates and delivery capacities is key in developing strategies to counter these problems.Master of Science in Chemical Engineering:
Knowledge about interactions between API’s and nanofibers allows to enhance our chemical technological knowledge, e.g. on the meso-scale regarding multi-scale design of polymer based processesMaster of Science in Sustainable Materials Engineering :
Design of nanofibers and their functional properties in order to enhance API delivery capacities within the body and increase the interactions between material and API.Master of Science in Textile Engineering:
Nanofibers of Poly(2-alkyl-oxazolines) provide access to bio-degradable nanoparticles which can be used in API delivery systems to counter the poor solubility of these pharmaceuticals. Design and production of nanofibers and the produced interactions with API’s are key in further development and use of these nanofibers in pharmaceutical industry.
- Study programmeMaster of Science in Biomedical Engineering [EMBIEN], Master of Science in Chemical Engineering [EMCHEM], Master of Science in Sustainable Materials Engineering [EMMAEN], Master of Science in Textile Engineering [EMTEXT]KeywordsPolymers, interactions, Nanoscale, modeling, active pharmaceuticalsReferences
(1) Goossens, H.; Catak, S.; Glassner, M.; de la Rosa, V. R.; Monnery, B. D.; De Proft, F.; Van Speybroeck, V.; Hoogenboom, R. ACS Macro Lett. 2013, 2 (8), 651–654.
(2) Bouten, P. J. M.; Hertsen, D.; Vergaelen, M.; Monnery, B. D.; Boerman, M. A.; Goossens, H.; Catak, S.; Van Hest, J. C. M.; Speybroeck, V. Van; Hoogenboom, R. Polym. Chem. 2014, 6, 514–518.
(3) Bouten, P. J. M.; Hertsen, D.; Vergaelen, M.; Monnery, B. D.; Catak, S.; van Hest, J. C. M.; Van Speybroeck, V.; Hoogenboom, R. J. Polym. Sci. Part A Polym. Chem. 2015, 53 (22), 2649–2661.
