Improved force-field approximations of symmetry-adapted perturbation theory
Improved force-field approximations of symmetry-adapted perturbation theory
Promotor(en): T. Verstraelen /25438 / Model and software developmentBackground and problem
Molecular dynamics is used in many scientific domains to simulate a system of interest at the atomic scale, e.g. "how does a drug molecule interact with an enzyme?" or "how is a gas adsorbed in a nanoporous material?" In these simulations, interatomic forces need to be computed very efficiently, e.g. for systems counting 104 to 107 atoms for 109 to 1012 time steps. In principle, one should solve the Schrödinger equation for the many-body electronic wavefunction in such a system, and then derive the forces acting on the nuclei, to perform a single molecular dynamics step. However, even approximate electronic structure theories are computationally far too demanding for the relevant time and length scales. Instead, force-field models are used, in which interatomic forces are modeled directly as a function of internuclear distances, without even trying to approximate the electronic wavefunction.
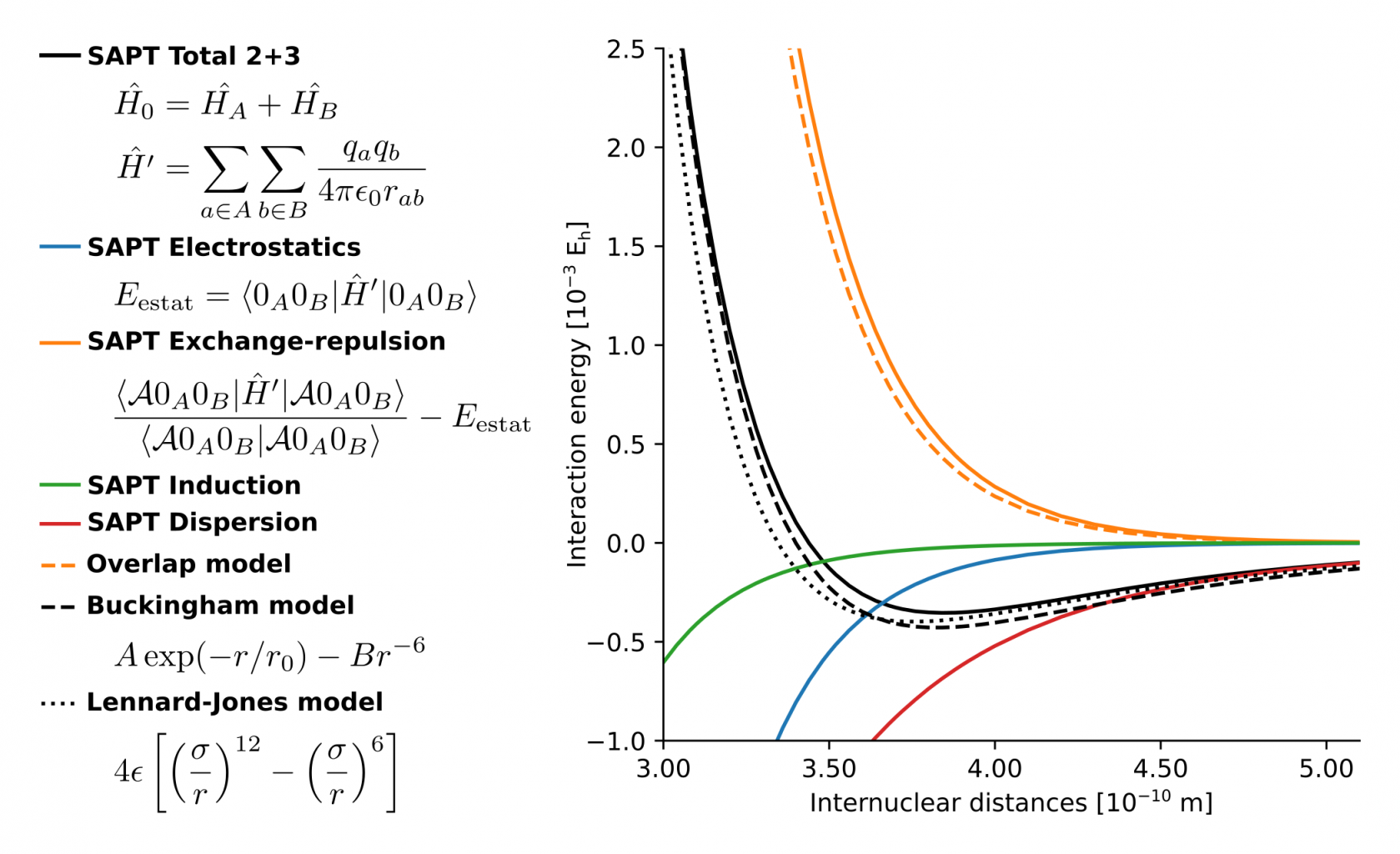
Figure 1. comparison of SAPT with some force field models for the argon dimer.
The simplest force field is the Lennard-Jones model, see Fig. 1, which is suitable for noble gases. More complex systems exhibit different types of interatomic forces, for which the functional form is not as trivial. Many of these models were proposed on empirical grounds, featuring a large number of fitting parameters, with little or no relation to the quantum-mechanical description of the electronic structure. This traditional approach is limited because one must estimate more empirical parameters for every new molecule or material of interest, which is a tedious and ambiguous task. Instead, it would be much more effective to derive force-field models directly from an electronic structure theory, using physically sound approximations.
Goal
We recently published a significant step in this direction, by proposing a force field for intermolecular interactions with only three adjustable parameters, the Monomer Electron Density Force Field (MEDFF). [1] The energy terms in MEDFF have a one-to-one correspondence with symmetry-adapted perturbation theory (SAPT) [2]. One of the zeroth-order terms in MEDFF is the so-called overlap model for short-range repulsion (due to Pauli exclusion) between two molecular fragments as follows:
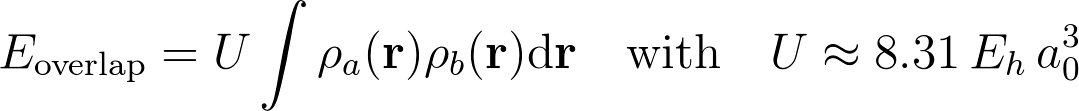
Where ρa and ρb are the electron densities of the isolated fragments. While this is a well-known model, there is no formal derivation, which could reveal the (system dependent) prefactor or which could lead to refinements of the overlap model.
The main goal of this thesis is to advance the overlap model by deriving it as an approximation of SAPT exchange-repulsion and by studying the impact of approximate densities in the overlap integral. A proper derivation will expose the assumptions of the current model at a theoretical level and will reveal potential avenues for improvements, e.g. by including terms that are currently neglected. At the moment, MEDFF uses a small number of exponential basis functions for the molecular electron density, which also introduces approximations. The impact of this approximation can by studied with numerical assessments. An efficient implementation of different versions of the overlap model will enable systematic validations, making use of the UGent HPC infrastructure. (See https://www.ugent.be/hpc) In first instance, the S66x8 dataset [3] will be used for validation. This is a standard benchmark comprising small molecular dimers whose intermolecular interactions have been studied extensively. In a later stage, models can be validated using condensed-phase simulations of liquids.
- Study programmeMaster of Science in Engineering Physics [EMPHYS], Master of Science in Physics and Astronomy [CMFYST]KeywordsMolecular simulation, Computational physics, intermolecular interactions, Force fieldsReferences
[1] S. Vandenbrande, M. Waroquier, V. Van Speybroeck & T. Verstraelen (2017) The Monomer Electron Density Force Field (MEDFF): A Physically Inspired Model for Noncovalent Interactions. Journal of Chemical Theory and Computation, 13(1), 161–179. https://doi.org/10.1021/acs.jctc.6b00969
[2] Stone, A. J. The Theory of Intermolecular Forces, Second edition.; Oxford University Press: Oxford, 2013.
[3] Řezáč, J., Riley, K. & Hobza, P. Extensions of the S66 Data Set: More Accurate Interaction Energies and Angular-Displaced Nonequilibrium Geometries. J. Chem. Theory Comput. 7, 3466–3470 (2011). http://dx.doi.org/10.1021/ct200523a.
